Notes on understanding cervical myelopathy
What does ‘degenerative’ mean?
‘Degenerative changes’ doesn’t just mean wearing away, it also means growth. Knobbly osteoarthritic knees are a good visible example of this apparent paradox.
The same thing happens in the cervical spine.
Just as skin dehydrates and sags with age, intervertebral discs dehydrate and flatten (1–3). As a disc flattens, the vertebrae above and below it press into each other more. “Settling like a pile of dishes”, as the great physiotherapist Gregory Grieve put it (4). The bony surfaces now bear more and more load.
In response, those bony surfaces thicken, much like skin forms calluses when overused. This new bone has a “brittle, sugary consistency”, as one early description from the 1950s puts it (5). It forms on facet joints, on uncovertebral joints, and extends into spurs on the posterior lips of vertebrae.
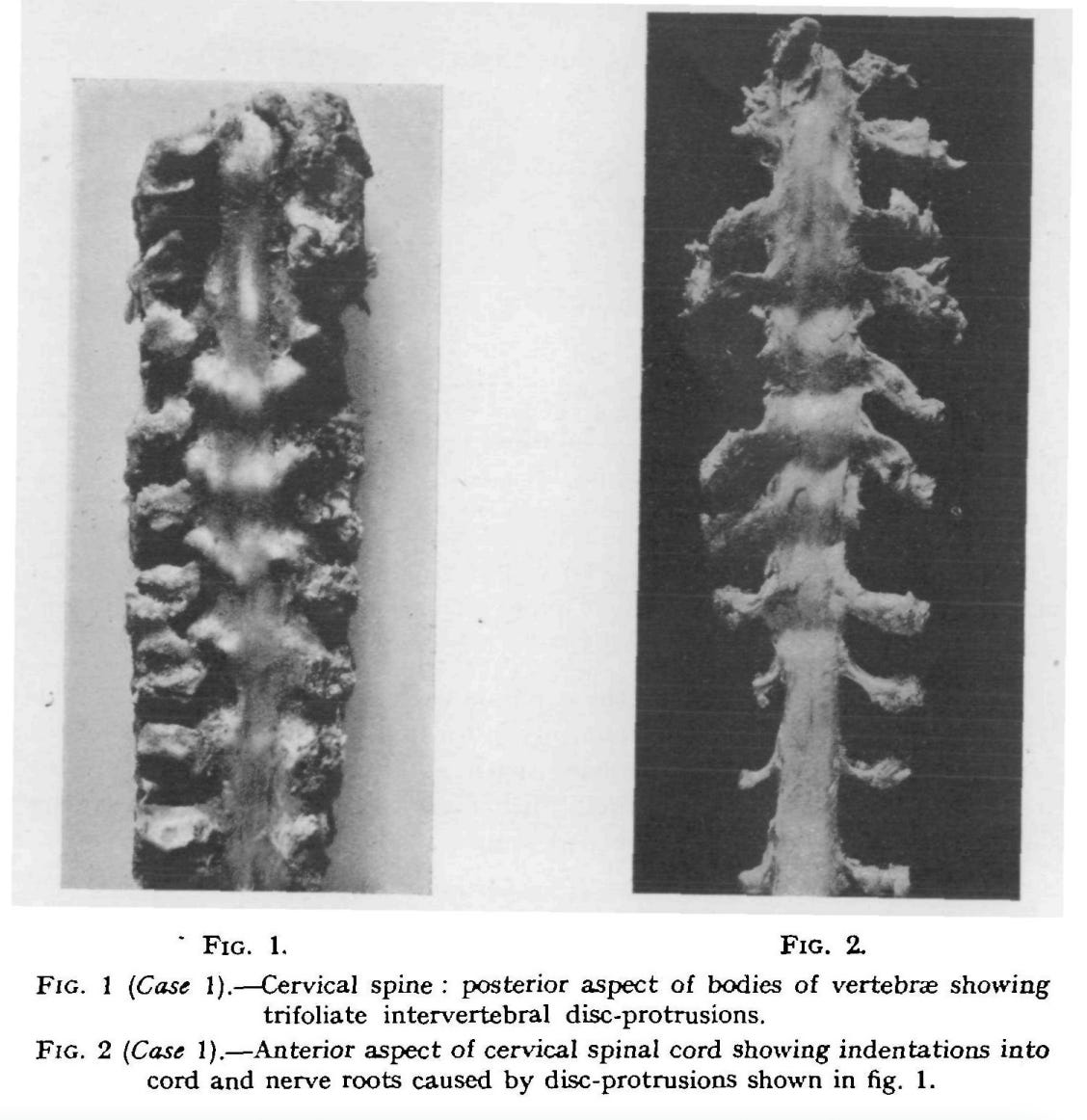
These bony extensions are useful in one sense: they support the spine against thinning discs. But they also crowd out the spinal cord. “At operation, when the dura is opened, the cord is found kinked and displaced backwards” (5).
Meanwhile, as discs flatten, the whole cervical spine shortens, and the already thickened ligamentum flavum slackens and buckles into the canal (2). Narrowing discs also bulge outwards.
So three kinds of structures end up encroaching on the cord:
bone, in the form of spurs and thickened joint surfaces
ligament, slack and buckling
disc, narrowed and bulging
The spine’s attempts to adapt to disc narrowing have become the cause of the problem. Yesterday’s solutions become today’s problems.
Mechanical stress on the cord
Bone, ligament and disc encroachment are usually described as compressing the cord. True, but these changes also add tension, shear, and even torsion (6). A bone spur pressing into the cord might cause myelopathy not where the cord is compressed, but on the opposite side where the cord stretches (7).
Neck movement adds dynamic stress on top of the static. When you flex your neck, the cord stretches over those degenerative changes. When you extend your neck, it gets pinched by them. Bone spurs create “deep grooves” in the cord’s anterior surface, says Alf Breig, forming “humps” on the posterior side (7).
This variety of stresses helps to explain why DCM varies so much:
between individuals
week to week in the same person, in the early stages
between limbs in the same person
It is also part of why no single test reliably detects it (8).
What happens to the cord?
All this mechanical stress squeezes blood vessels and chokes the cord’s blood supply (2,8). The first surgeons to document DCM described how the cord blanched under stress points (5,9). “On flexing the neck there was posterior displacement of the cord, producing pallor” (5), wrote one.
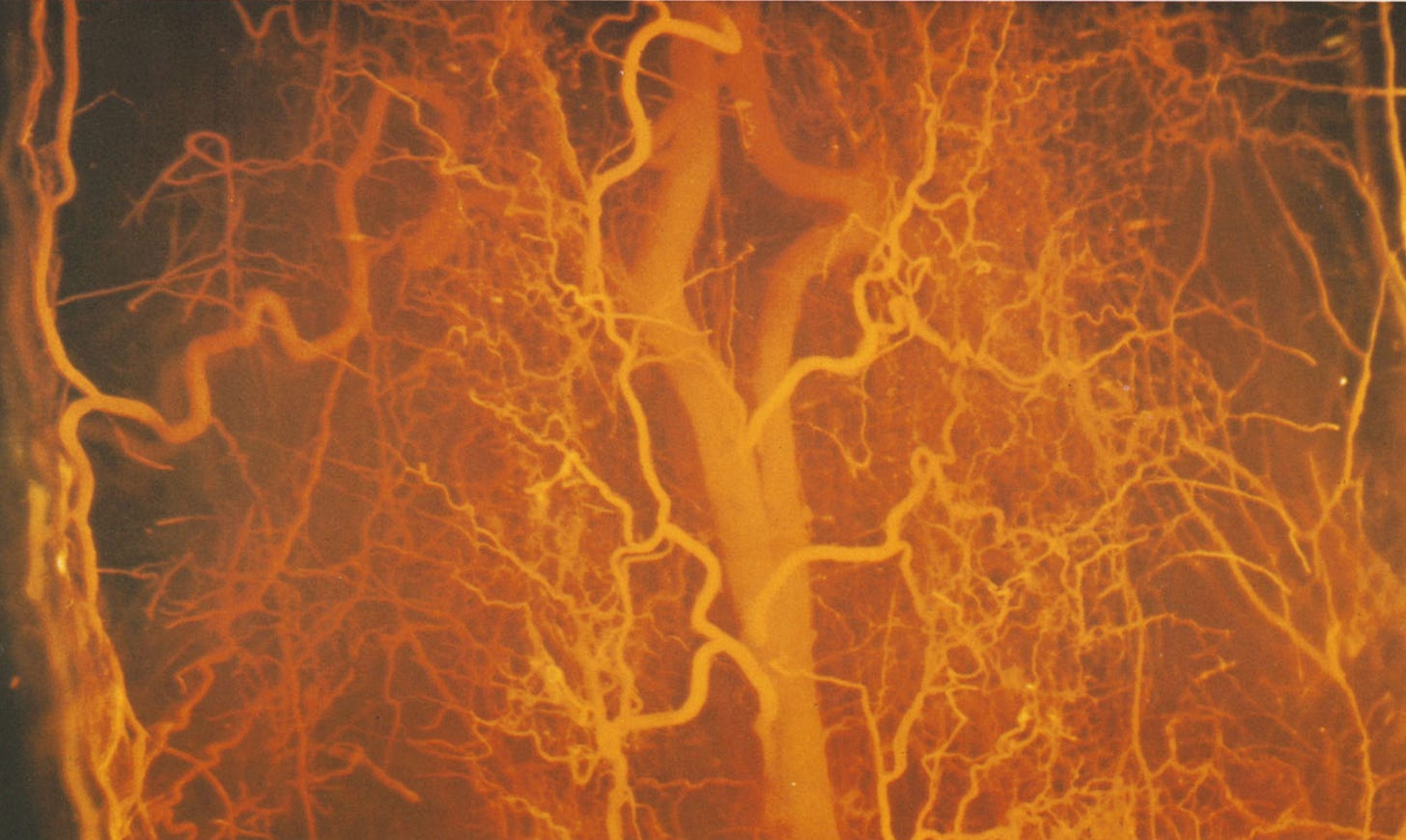
Starved of blood, neurons lack oxygen and can’t fire. It’s similar to sitting with your legs crossed for too long, reducing blood flow to your peroneal nerve and giving yourself a temporary numb, floppy foot.
With DCM, blood vessel walls grow thicker to adapt to the chronic mechanical stress. But in doing so, the space inside the vessel shrinks. So even as blood vessels try to adapt, the cord is starved for oxygen.
Eventually, lacking oxygen and surrounded by inflammation from a damaged blood-spinal cord barrier — through which fluid and inflammatory cells leak into the cord tissue itself — neurons and their support cells die (6,8).
A paradox: weakness and overactivity
Upper motor neurons do two jobs at once. They drive movement, and they control it: tapping the brakes to inhibit muscle contractions, dampen reflexes, and relax tone. When UMNs stop working, you lose both the drive and the brakes. So in DCM, the legs are weak and in a constant state of agitation.
This is why we see jumpy reflexes. From just a light tap, the knees jerk dramatically. The calf muscles may contract rhythmically — clonus — because the reflex is so sensitive that it elicits itself over and over in a cycle. When the sole of the foot is scratched, the big toe curls upwards: Babinski’s sign, normally seen only in neonates, re-emerges when corticospinal inhibition is lost (12).
Combine the hypertonia, hyperreflexia and the weakness, and you get myelopathic gait (13–15). It’s often:
short-stepping: weakness shortens stride
stiff: hypertonia
slow: weakness, again
shuffling: less toe clearance, what one author calls a “spastic foot drop” (13). Falls are often the first reason the myelopathic patient presents to the health system
strenuous: every step is work
Numbness in the legs makes everything worse.
Patients say:
“My legs are like jelly.”
“It’s like I’ve had too many pints!”
“I keep wandering to one side.”
“I’m dragging my legs.”
“I’ve had to start using a stick...”
The same paradox in the arms
When upper motor neurons in the neck become damaged, the arms develop the same pattern: simultaneous rigidity and jumpy reflexes from lost inhibition, weakness from lost drive.
A light flick on one digit elicits a reflex in another: Hoffmann’s and Tromner’s signs. Movement becomes stiff, slow and strenuous. Doing up buttons is a classic difficulty.
In clinic, ask your patient to:
rapidly open and close a fist
hold and extend the fingers
tap a finger (9,16,18)
Patients say:
“My arms feel dull.”
“I can’t feel my fingertips.”
“I can’t use a knife and fork like I used to, I’m getting so clumsy.”
“My hands shake.”
To compensate for lost dexterity, larger joints — wrist, elbow and shoulder — take over movements that should require only fine motor control (19).
The arms differ from the legs in one important way, though. For the legs, the cervical cord is just a highway: upper motor neurons passing through. For the arms, the cord is also a nerve root entry/exit point.
Lower motor neuron cell bodies sit in the ventral horn and their axons exit through the ventral roots, while sensory axons enter through the dorsal roots from their cell bodies in the dorsal root ganglia just outside the cord. So cervical degeneration in the arms can also cause nerve root problems: radiculopathy.
Radiculopathy is different to myelopathy because there’s no hyperactivity. It’s a more straightforward loss of function: weakness, decreased reflexes.
Which means, paradoxically, that at the actual level of degeneration, a patient with DCM might have none of the expected hyperreflexia or hypertonia. There’s a radiculopathy, with its pure loss of function, at the myelopathy point.
Nerve root symptoms
With nerve root injury comes nerve root symptoms. A few are worth keeping in mind:
Transient tingling in the hands, often with numbness. Frequently misdiagnosed as carpal tunnel syndrome (3).
Scapular pain (20): pain referred from the spine to the shoulder blade, commonly misattributed to local muscle problems like trigger points or rhomboid strains. Similar to how spine-referred buttock pain gets mislabelled as gluteal or piriformis issues.
Radicular pain: distal, severe, neuropathic. The worst of the nerve root symptoms. But unlike disc herniations, where inflammation drives immediate pain, DCM doesn’t always produce radicular symptoms. Tingling is more common.
And what about neck pain? It’s common, as it is for all older adults. But it’s often not as serious as you might expect. “It is rare for pain, stiffness, or limitation of movement of the neck to be an outstanding symptom”, one doctor wrote in 1955 (21). “Examination of the neck commonly reveals little abnormality.”
A normal neck assessment does not exclude DCM.
The atypical
Even this is only the shallow end. Patients also report:
sensory disturbances — “a wet gel substance”, says one (22)
sciatica (23)
altered temperature sensation
nausea and vomiting
difficulty breathing
cranial nerve symptoms: difficulty swallowing, dizziness, headache, tinnitus, face pain and numbness (20)
These symptoms might be downstream effects in the brain, or autonomic dysfunction. Or they might arise because the blood vessels supplying the cord criss-cross spinal levels, so a single local stress point can starve a much wider region of oxygen (8).
“There is less correlation than might be expected between the distribution of the [myelopathic signs] and the site of the lesion”, wrote Walter Russell Brain in 1952, in one of the first papers to describe DCM (24).
At least we have a decent understanding of the basics.
***
Thanks to John Drummond for his help with this post. A different version appeared in MSK Mag.
References
Nouri A, Tetreault L, Singh A, Karadimas SK, Fehlings MG. Degenerative Cervical Myelopathy: Epidemiology, Genetics, and Pathogenesis. Spine. 2015 Jun;40(12):E675–93.
Tetreault L, Goldstein CL, Arnold P, Harrop J, Hilibrand A, Nouri A, et al. Degenerative Cervical Myelopathy: A Spectrum of Related Disorders Affecting the Aging Spine. Neurosurgery. 2015 Oct;77(Supplement 1):S51–67.
Clarke E, Robinson PK. Cervical myelopathy: a complication of cervical spondylosis. Brain. 1956;79(3):483–510.
Grieve GP. Common vertebral joint problems. Edinburgh: Churchill Livingstone; 1981.
Allen KL. Neuropathies caused by bony spurs in the cervical spine with special reference to surgical treatment. J Neurol Neurosurg Psychiatry. 1952 Feb;15(1):20–36.
Davies BM, Mowforth OD, Smith EK, Kotter MR. Degenerative cervical myelopathy. BMJ. 2018 Feb 22;k186.
Breig A, Turnbull I, Hassler O. Effects of mechanical stresses on the spinal cord in cervical spondylosis: a study on fresh cadaver material. J Neurosurg. 1966 Jul;25(1):45–56.
Badhiwala JH, Ahuja CS, Akbar MA, Witiw CD, Nassiri F, Furlan JC, et al. Degenerative cervical myelopathy — update and future directions. Nat Rev Neurol. 2020 Feb;16(2):108–24.
Crandall PH, Batzdorf U. Cervical spondylotic myelopathy. J Neurosurg. 1966 Jul;25(1):57–66.
Karadimas SK, Gatzounis G, Fehlings MG. Pathobiology of cervical spondylotic myelopathy. Eur Spine J. 2015 Apr;24(S2):132–8.
Lees F, Turner JWA. Natural history and prognosis of cervical spondylosis. Br Med J. 1963 Dec 28;2(5373):1607–10.
van Gijn J. The Babinski sign: the first hundred years. J Neurol. 1996;243(10):675–83.
Rubino FA. Gait disorders. The Neurologist. 2002 Jul;8(4):254–62.
Maezawa Y, Uchida K, Baba H. Gait analysis of spastic walking in patients with cervical compressive myelopathy. J Orthop Sci. 2001;6(5):378–84.
Pirker W, Katzenschlager R. Gait disorders in adults and the elderly. Wien Klin Wochenschr. 2017;129(3):81–95.
Smith ZA, Barry AJ, Paliwal M, Hopkins BS, Cantrell D, Dhaher Y. Assessing hand dysfunction in cervical spondylotic myelopathy. PLOS ONE. 2019 Oct 28;14(10):e0223009.
Good DC, Couch JR, Wacaser L. “Numb, clumsy hands” and high cervical spondylosis. Surg Neurol. 1984 Sep;22(3):285–91.
Miwa T, Hosono N, Mukai Y, Makino T, Kandori A, Fuji T. Finger-tapping motion analysis in cervical myelopathy by magnetic-sensor tapping device. J Spinal Disord Tech. 2013 Aug;26(6):E204–208.
Durrant DH, Blum JW, True JM. Myelopathy, radiculopathy, and peripheral entrapment syndromes. Boca Raton: CRC Press; 2002.
Munro CF, Yurac R, Moritz ZC, Fehlings MG, Rodrigues-Pinto R, Milligan J, et al. Targeting earlier diagnosis: what symptoms come first in Degenerative Cervical Myelopathy? PLOS ONE. 2023 Mar 31;18(3):e0281856.
Northfield DWC. Diagnosis and treatment of myelopathy due to cervical spondylosis. Br Med J. 1955 Dec 17;2(4954):1474–7.
Mowforth OD, Davies BM, Kotter MR. “I am not delusional!” Sensory dysaesthesia secondary to degenerative cervical myelopathy. BMJ Case Rep. 2019 Apr 1;12(4):e229033.
Han C, Wang J, Wang L, Gong Q, Huang W. Sciatica-like pain caused by cervical spondylotic myelopathy: four case reports and systematic review. Front Med. 2024 Aug 1;11:1429618.
Brain WR, Northfield D, Wilkinson M. The neurological manifestations of cervical spondylosis. Brain. 1952;75(2):187–225.


Thank you for this, another level of insight.